Many people still keep radio components of the USSR times at home. They have silver contacts that can be removed at home. To get the precious metal, you will need to study the procedure and prepare special items.


Features
The contacts of electrical components may contain pure silver. There are several types of connections.
- Those that do not magnetize. The silver content in this group is the highest.
- Magnetizable. They have the least amount of pure metals.
- Copper. Soldering is a copper plate, which is covered with a small layer of silver.
Silver contacts are used in devices with low power. Pure metal can be mined from:
- electromagnetic starters;
- electric machines;
- relay;
- temperature sensors;
- silver-zinc batteries.


Care Rules
Silver contacts need to be monitored. Care consists in periodically checking their condition. If they are dirty, try cleaning with a suede or cloth.. It will need to be slightly moistened in gasoline. Can be cleaned with a stiff hair brush.
The file removes the influx.


How to separate silver?
It has long been practiced to extract pure silver from contact alloys. To produce pure metal, special purification techniques called refining are required. This procedure is divided into several steps, the result of which is always the selection of silver.
Although refining is an industrial procedure, you can solder the precious metal yourself at home. There are different ways.

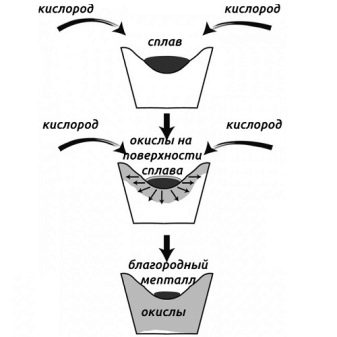
Cupellation
Silver can be extracted from base alloys by cupellation.This method is based on the ability of molten silver with lead to begin to oxidize in air. With such contact, the metal will separate. To work, you will need a special furnace equipped with a crucible in the form of a bowl. The bowl should be with a marl coating based on porous limestone clay.
The process is divided into several stages:
- the oven is warming up;
- then a test crucible is placed in it, in which technical silver and lead already lie;
- warm the crucible until a full melting occurs;
- after that, air masses are launched into the furnace.
A thermal reaction is included in the process. When it ends, you can pull out the crucible and pour its contents into forms.
Cooling, the alloy takes on a rainbow of colors. Similar reports that, in addition to silver, there are other precious metals in its composition.

Electrolytic method
To extract metal in this way, special plastic or sandstone cells with silver nitrate will be required. The precious metal content in the liquid is at least 50 g / liter. Contaminated silver will be used as the anode. The cathode in this situation is a stainless steel cut into strips of small thickness.
Anodes are placed in textile bags. Contaminated silver particles that could not dissolve will then be collected in these bags. At the cathodes, silver macrocrystals will be deposited. They will grow towards the opposite pole to short circuit. To prevent shorting, the branches of the crystals are broken during mixing of the solution. Such crystals independently settle on the bottom, from where they are removed. Directly from these components and you can melt the ingots.

Chemical
The extraction of silver metal from solutions or salt will be carried out using a chemical method. As a result, black silver sulfate will be obtained. To use this method, mandatory use of sodium sulfate will be required. The whole procedure will last until silver sulfate ceases to be released.
If this method is chosen, the precious metal is obtained only in the form of chloride and only after the addition of a certain substance. You can use ammonium chloride or sodium chloride. The result is a liquid that must settle until it is divided into two parts: cloudy and clean.
Advice! If in the future salt is added and the solution does not cloud, then the entire precious metal has settled on the bottom.

If you need to work with chloride, there are two methods:
- casting - using alkali metal carbonate;
- using a solution - you will need to bring the sample to the highest values.
Each of the described methods can be carried out independently at home. For work, special substances and tools will be required. The easiest method for people who do not have refining experience is electrolytic.
It is divided into three stages:
- precious metal is initially dissolved in nitric acid;
- then it is cemented;
- the final stage is fusion.


In order to carry out the first stage, you will need to take a solution of nitric acid, the concentration of which is 68.8%. Together with this, you need to prepare a glass container and a quartz wand. When working with acid, you must follow safety precautions and be in a well-ventilated area. The best option is outdoor interaction.. The skin of the hands should be protected with gloves, and the eyes should be covered with glasses. Advice! It should be remembered that acid is poured into water. Pouring water into acid is prohibited.
To dilute nitric acid, you need to take deionized water and pure acid. It is necessary to observe a ratio of 1: 1. The formed liquid is mixed with a quartz wand.Further, the acid is poured into glass containers intended for reagents. Silver nitrate should be produced so that it is enough for the whole procedure. The approximate concentration is 50 grams per liter. Now you need to dissolve the silver in a liquid. Usually, during this procedure, NO2 is released, after which the solution acquires a blue tint. Immediately you need to be prepared for the fact that the procedure requires a large amount of time. Entirely precious metal will dissolve only after 10 hours. The formed solution is closed in a can for chemical reagents.


The next step is cementing silver. It will require the separation of metallic silver from nitrate, in which copper is present. To do this, add copper to silver nitrate. You can use old water pipes that have previously been cleaned to a shine. When copper is added, the reaction accelerates. As a result, cement silver forms on the tubes, which looks like powder. So that the reaction rate does not decrease, it will be necessary to periodically remove the cement from the tubes into the solution.
The whole process becomes possible due to the fact that the tubes transfer copper to silver nitrate, which gradually leads to complete dissolution. If the tubes have disappeared, they must be replaced. As silver is supplanted, the reaction begins to slow down. For this reason, you can not follow the process and leave it for a couple of days.
All that is needed is to observe the presence of copper and to ensure that no excess objects are present in the solution.


It is possible to determine that the reaction is complete by cooling the solution, in which there are no signs of the current process: at the top, the liquid will acquire a blue and pure color, and cement will be located below. When the reaction is complete, you will need to filter the cement.
To work, you need to prepare:
- funnel;
- coffee filters;
- capacity where raw materials are removed.
Advice! It must be remembered that filtering is carried out repeatedly. At least five procedures are required.



This approach will remove all particles of copper nitrate from cement. Upon completion of the filtration, you need to evaporate the excess moisture or wait until it is removed naturally.
In the next video, you will be able to clean technical silver at home.










